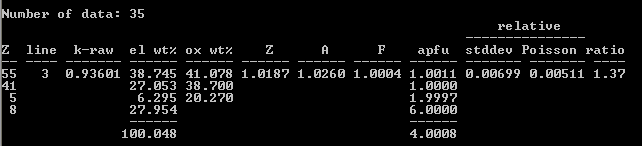
An advantage of having two potential standard materials for a given element is the ability to cross-check them; this is made especially convenient when the matrix corrections are small. When I analyze my most recent batch of CsNbOB
2O
5 for Cs using my most recent batch of CsTiOAsO
4 as the standard, I get the results below when analyzing 35 spots. I’ve fixed wt% Nb
2O
5 and wt% B
2O
3 at the respective stoichiometric values. Likewise, I’ve assumed that the CsTiOAsO
4 is stoichiometric.
PAP/MAC30:
Armstrong/FFAST:

The small difference in atomic number correction between the two models accounts fully for the small difference in results. Further, the results suggest that both compounds are pure and stoichiometric or very nearly so.
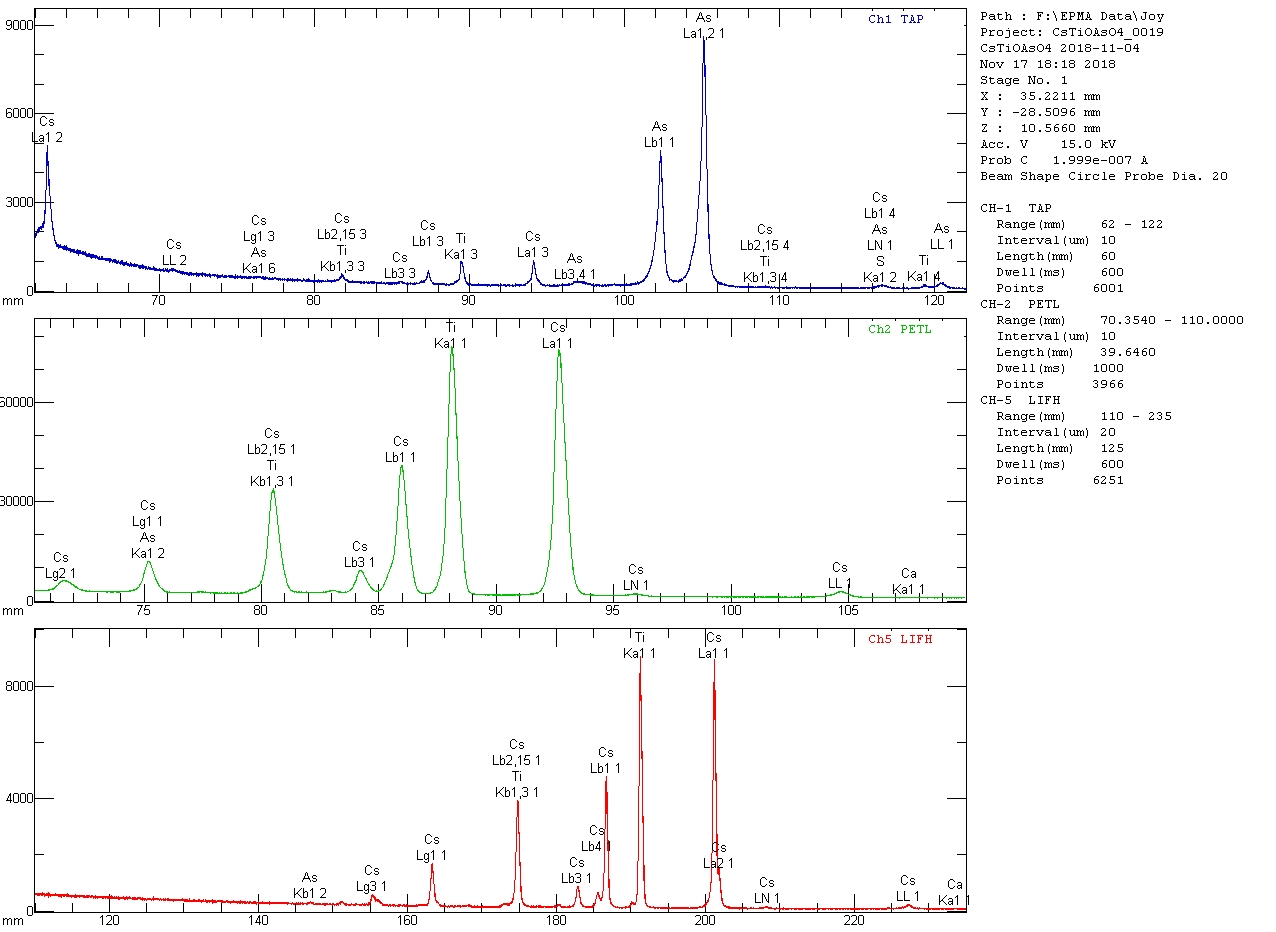
EDIT: I just completed a set of wavelength scans on my most recent batch of CsTiOAsO
4, and they reveal a minute Ca Kα peak corresponding to less than 500 ppm CaO. I'd noted the presence of Ca in the remnants of the flux that got into the mount; I'm not sure where it came from, but it doesn't appear to have contaminated the CTA significantly. The small peak near 151 mm on LiFH is Cs Lγ
4. I'm not certain of the identity of the peak at 180.3 mm on LiFH and at 83.05 mm on PETL (it also shows up on a scan on a CTA crystal from another batch); it is not Zn Kβ(2) and is likely to be a minor Cs peak, perhaps Cs Lβ
6.