I decided to draw up a couple of simple schematic diagrams to help explain the Bragg defocus effect on secondary fluorescence from boundaries.
Let's take the impressive example of measuring Cr in a basaltic glass adjacent to a chromium phase, for example Cr2O3 or chromite, as seen here:

Because the Fe x-rays emitted from the beam spot in a basaltic glass easily fluoresce the Cr K shell in the adjacent oxide, we get almost 1 wt.% of SF artifact at 1 um from the boundary and even at 100 um from the boundary we still should measure ~100 PPM of Cr in the glass,
even if there is no Cr in the glass at all!
Now, there is a strong orientation effect, though *not* for EDS. With an EDS detector we will see essentially the secondary fluorescence artifact we have modeled because EDS detectors show no collimation effects for several millimeters or more from the beam position (hence the origin of so-called "system" peaks).
And for WDS spectrometers where the glass to oxide boundary is oriented parallel to the length of the WDS Bragg crystal there is also minimal defocusing effect, therefore what the Standard SF model shows is what one will observe in actual measurements. This is the situation shown in this schematic:
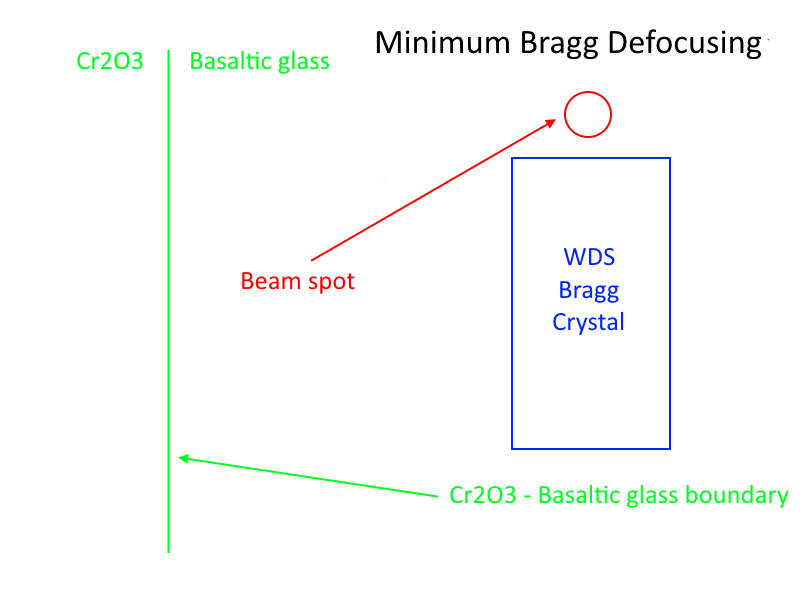
However, if the glass to oxide interface is oriented perpendicular to the length of the WDS Bragg crystal we will have a strong defocusing effect, and the actual secondary fluorescence artifact from the adjacent oxide phase will be decreased depending on the spectrometer geometry details (spectral resolution, sin theta, etc). This is the situation shown here:

And for "in between" spectrometer to boundary orientations, the degree of Bragg defocusing is also "in-between"!

My next project will be to start devising a procedure to characterize the degree of Bragg defocusing for one's WDS spectrometers, in order to provide a defocus correction to the secondary fluorescence from boundary phases correction in the matrix correction! A correction of a correction of a correction!
Please let me know if I didn't explain this clearly enough. It's quite complicated.
john