Maybe this would be a good time to examine the method of Mean Atomic Number (MAN) background correction in more detail.
The example I am utilizing is from Paul Carpenter at Washington University (used with permission) and Paul acquired this MAN calibration data set for use in quantitative x-ray mapping with Probe Image and CalcImage as can be seen in the beautiful quant x-ray maps attached below. Note the stoichiometric *and* total quant maps which can be automatically generated and output with CalcImage.
Of course the MAN background correction is good to a few hundred PPM in silicates and oxides so it really is "overkill" for quant x-ray mapping, but it sure does save a lot of time because one doesn't need to acquire any off-peak x-ray maps!
So, what is the basis for the the MAN correction? The details are available in this publication:
http://probesoftware.com/Improved%20MAN%20%28Jour.%20Micros.%20Microa.,%201996%29.pdfbut it suffices to say that it is based on Kramer's Law, where in the absence of the element under investigation, the on-peak x-ray production of the material can be used to calibrate the continuum background. The MAN correction also contains an adjustment for absorption of the continuum of the emission line being investigated and for the matrix in question (both for the MAN calibration standards and the unknowns). Since the standard compositions are already known, the calculation of mean atomic number (MAN) is easily performed. In the case of unknowns, the mean atomic number of the sample is calculated during the ZAF or phi-rho-z matrix iteration.
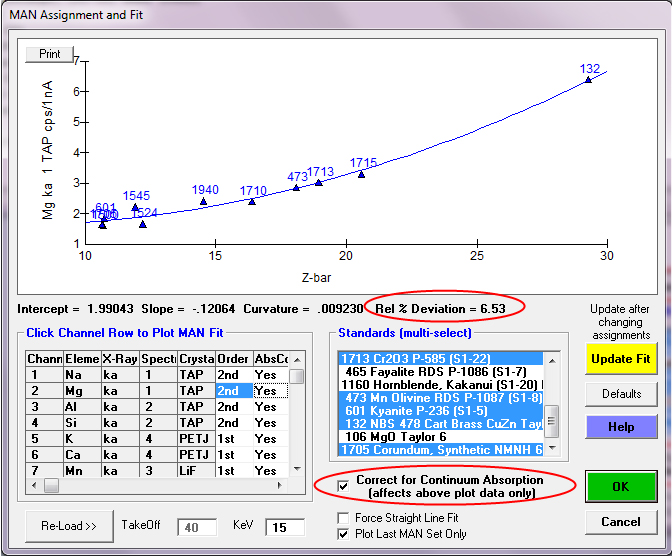
So now let us examine the first element measured in these x-ray maps, Na ka as seen here in the Analytical | Assign MAN Fits menu available in both Probe for EPMA and CalcImage:
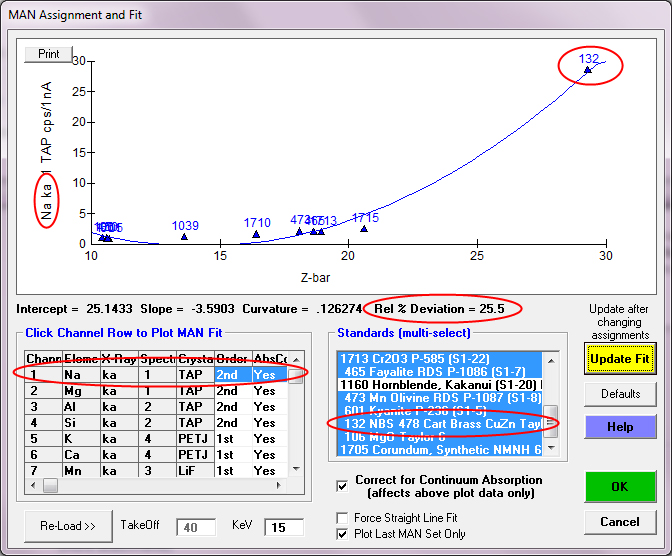
Of course, we can immediately see one problem, and that is that the NBS CuZn alloy is very much an outlier in our Na MAN fit. Why is that? Well it's because of our old friend, the Zn La on Na interference. Now, this interference could be corrected for in PFE- but let's not get carried away! There are plenty of other MAN standards to fit for Na, and besides, we aren't measuring materials with that high an average Z anyway!
So let's unselect that standard from our MAN fit by using a <ctrl> click of the mouse and then we click the Update Fit button to re-plot the fit as seen here.
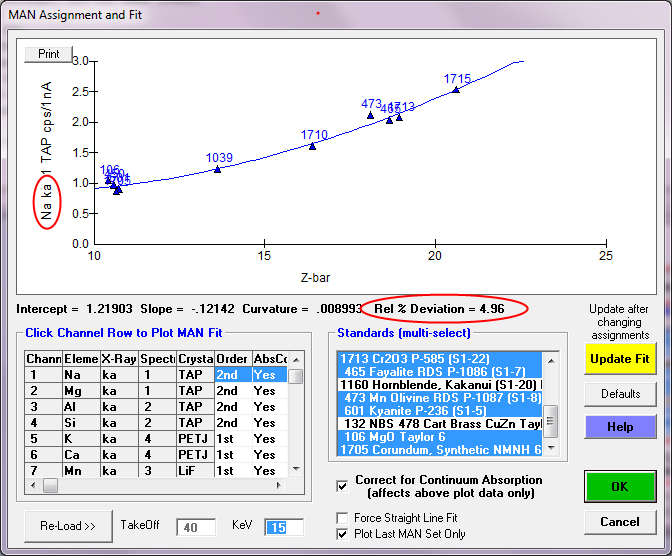
Note the now much more reasonable fit to the on-peak x-ray intensities from our standards, which is loaded by default based on whether the standard contains the element under investigation. Since the standard composition database indicated no Na in the NBS CuZn alloy, the software loaded that standard intensity. But for other standards that actually contain Na, for example, Kakanui Hornblende, the x-ray intensities for that standard material are not loaded for the default MAN fit.
To demonstrate the necessity of the continuum absorption correction, we can unselect the correction for continuum absorption by unchecking the checkbox and the significantly worse fit is quite evident. Why is this? Because every standard composition absorbs continuum radiation (i.e., at the characteristic energy of our elements of interest) in somewhat different degrees, depending on the physics details- that is why we have absorption edges in our EDS continuum spectra!
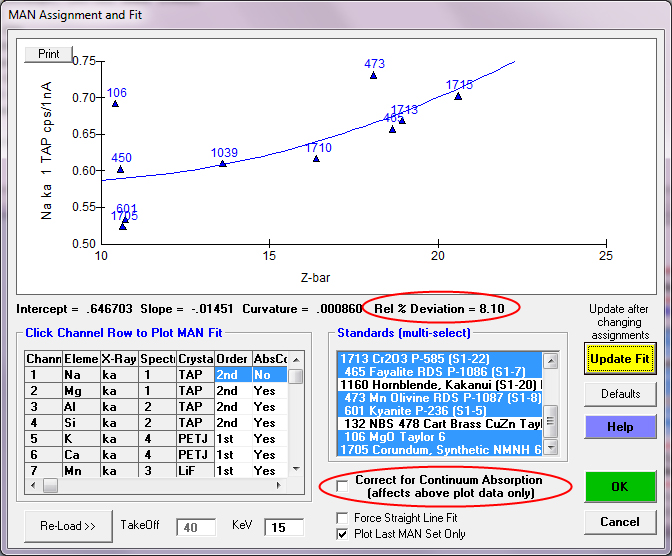
So clearly the correction for continuum absorption is necessary, as seen also in this plot of Mg ka in standards that do *not* contain Mg:
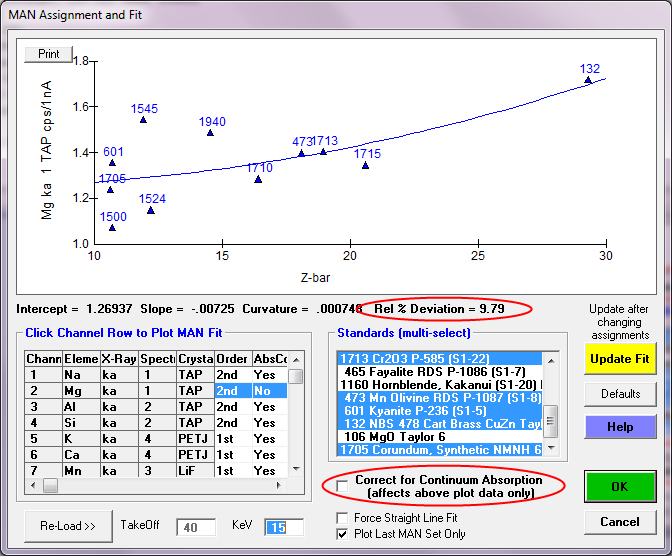
By including the correction for continuum absorption for Mg ka we are back in business as seen here:
Don't worry, the program defaults all these options automatically!

So, let's move on to a different situation as seen here for Mn ka:
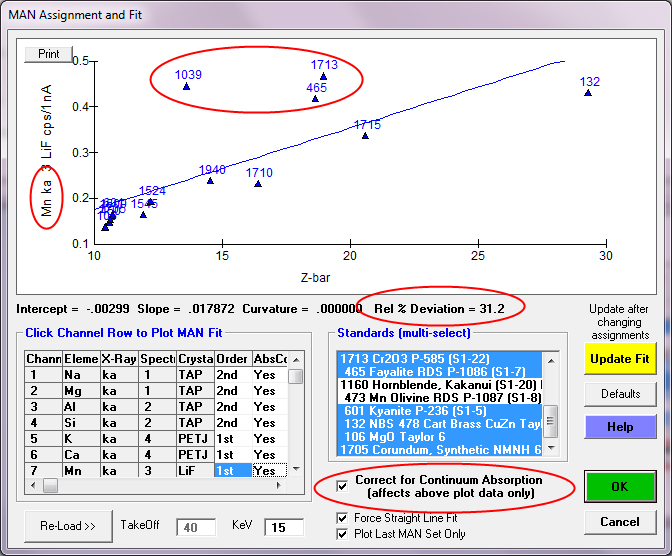
Here, again, we see some outliers in our MAN fit, but these are *not* from spectral interferences from other elements, as was the case for Zn La on Na. These are standards which contain small amounts of Mn that have *not* been specified in the standard composition database. Paul puts it himself this way:
"1039 is our wollastonite standard that contains a low concentration of Mn but has not been characterized in the standard analysis yet. 1713 is Elba hematite that is also being adjusted, and the synthetic fayalite 465 also contains very low Mn. On a wavelength scan this level of Mn is not apparent."
By simply unselecting these standards which contain trace amounts of Mn, we can get a more accurate model of the background for our Mn measurements. Remember: background is by definition the lowest intensity one can measure! Interferences and "contamination" will all cause elevated outliers to the fit. Of course there are some subtle continuum artifacts as documented here:
http://probesoftware.com/Ti%20in%20Quartz,%20Am.%20Min.%20Donovan,%202011.pdfbut those are in the sub 100 PPM level! So not exactly significant for quant-xray mapping even when measuring off-peak images!
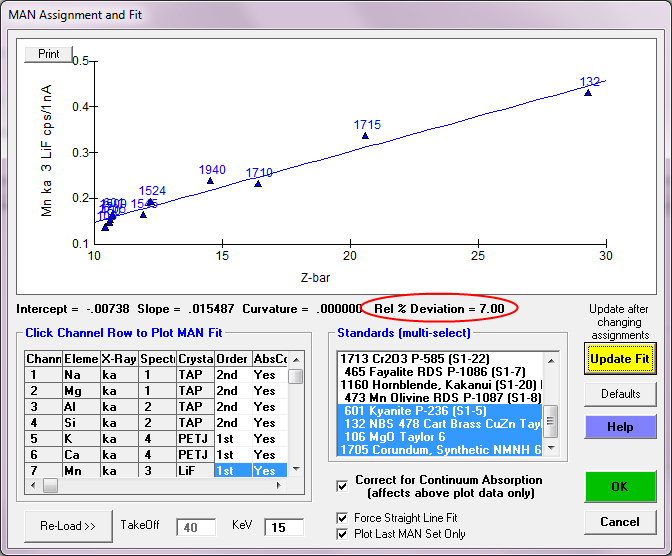
So after removing those standards from our fit which contain "undocumented" Mn, we obtain the following fit:
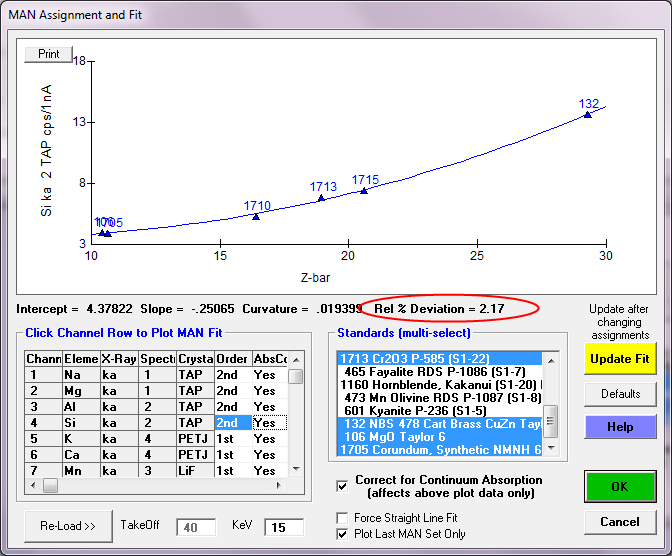
For an element such as Si or Al we obtain beautiful fits using the default MAN assignments made automatically by the program as seen here:
Now check out the gorgeous quant x-ray maps Paul acquired which are automatically corrected for background, matrix and spectral interferences in the attachments below!
It should also be mentioned that at zero concentration levels, using MAN background acquisition improves measurement precision by roughly the Sqr(2) and at the same time cuts the necessary acquisition time by 50%.
See the attachments posted here for details:
http://probesoftware.com/smf/index.php?topic=29.msg237#msg237